Consequences of Dysphagia
Understanding serious problem about failing to adhere to treatment.
Dysphagia describes eating, drinking and swallowing difficulties in infants, children and adults. People with dysphagia often have other health conditions that they are being treated for, which can affect their eating, drinking and swallowing abilities. .[1]
In one study, one in eleven primary care patients had frequent difficulties in swallowing tablets and capsules while GPs grossly underestimated these problems. Therefore, health care professionals should rule out swallowing difficulties regularly to avoid non-adherence and inappropriate drug modifications. Special attention should be paid to specific patient groups (e.g. women and patients with dysphagia, dysphagia indicators, or mental illness).[2]
Adherence to therapies is a primary determinant of treatment success. Failure to adhere to treatment is a serious problem which not only affects the patient but also the health care system. Medication non-adherence in patients may lead to substantial worsening of disease, death in some cases and increased health care costs. Barriers to adherence could be addressed as patient, provider and health system factors, with interactions among them. Identifying specific barriers for each patient and adopting suitable techniques to overcome them will be necessary and Healthcare professionals such as doctors, pharmacists and nurses have a significant role in their daily practice to improve patient medication adherence.[3]
Clinical Evaluation of oesophageal dysphagia in the elderly[4]
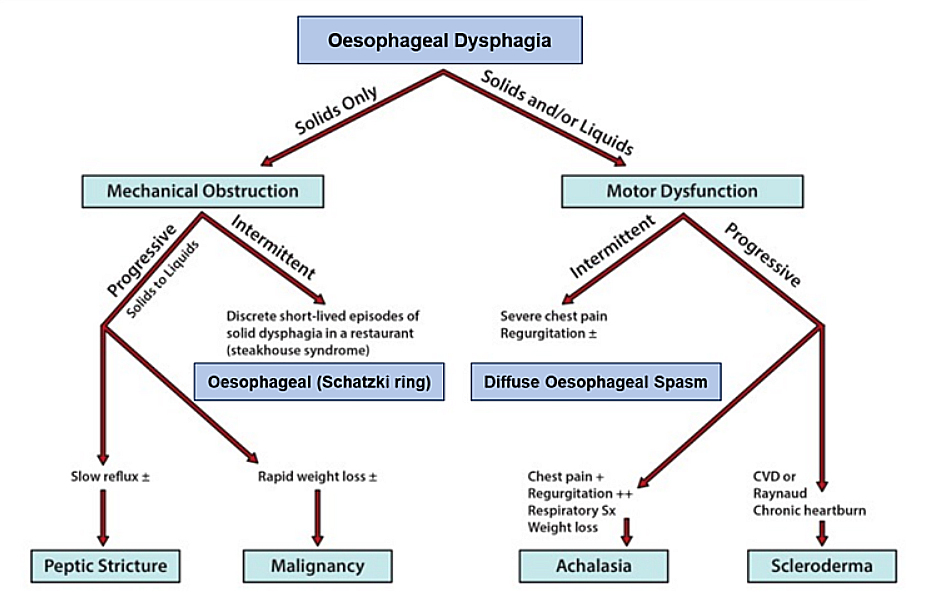
Elderly patients are particularly at risk of developing drug-related dysphagia and pill oesophagitis. They consume more medications and are more likely to have anatomic and motility abnormalities, cardiac enlargement with concomitant compression of the mid-oesophagus, and decreased saliva production. Thus, a detailed review of medications and when they are taken is an important component of dysphagia evaluation.[4]
Tablet crushing
Solid oral dosage forms such as tablets and capsules are the most common formulation for medications. Unfortunately, many people struggle to swallow solid oral dosage forms whole and instead cut or crush tablets or open capsules to make them easier to swallow. People who have dysphagia, find it necessary to modify their solid dose medications. Members of the general public who are otherwise healthy and able to swallow food and liquids may also crush their tablets because they perceive them to be too large or difficult to swallow whole. The proportion of the population that modifies their tablets in different settings varies, depending on the survey methods, from around 10–20% in the general population to 18–80% in aged-care home facilities and hospitals. [5]
Pharmacological Dysfunction
Crushing tablets or opening capsules can impact on the efficacy and safety of a medicine. When it disrupts a drug’s sustained-release properties, the active ingredient is no longer released and absorbed gradually, resulting in overdose. When a gastro-resistant layer is destroyed by crushing, underdosing is likely. The active ingredient released may degrade on contact with light, moisture or the food with which it is mixed for administration. The person who crushes the tablets or opens the capsules is exposed to drug particles, which may be carcinogenic, teratogenic or fetotoxic. They are sometimes allergenic. In practice, there are many drugs that should never be crushed or opened. Before crushing a tablet or opening a capsule, it is better to consider and research the impact it will have on the drug’s effects. It is sometimes preferable to use a different dosage form, or a different active ingredient.[6]
ROS000051-001 DOP September 2025
- Dysphagia and eating, drinking and swallowing needs overview. Available at: https://www.rcslt.org/speech-and-language-therapy/clinical-information/dysphagia/ (Last accessed Aug 2025)
- Difficulties swallowing solid oral dosage forms in a general practice population: prevalence, causes, and relationship to dosage forms. Available at: https://pubmed.ncbi.nlm.nih.gov/23052416/ (Last accessed Sep 2025)
- Patient Medication Adherence: Measures in Daily Practice. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3191684/ (Last accessed Sep 2025)
- Dysphagia in the Elderly. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3999993/ (Last accessed Sep 2025)
- Perceived Usability of Tablet Crushers: Comparison of Devices by People with and without Limited Hand Functions. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9961238/ (Last accessed Sep 2025)
- Crushing tablets or opening capsules: many uncertainties, some established dangers. Available at: https://pubmed.ncbi.nlm.nih.gov/25325120. (Last accessed Sep 2025)




